What is it?
Uracil is one of the four nucleobases in the nucleic acid of RNA. It is represented as the letter "U" when bonding with other bases in RNA. In RNA Uracil plays an important role, and binds to another base in RNA Adenine or "A". Even though Uracil is found in RNA, it is replaced by another nucleobases called Thymine or "T" in DNA.
As a pyrimidine nucleobase, Uracil is considered to be a heterocyclic aromatic organic compound. A heterocyclic compound is a cyclic compound that contains atoms of at least two different elements as a member of its ring or rings. The opposite of a heterocyclic compound is a homocyclic compound, in which the ring or rings is made of a single element. Since Uracil is a heterocyclic compound it contains different elements on it's ring, those elements are; carbon and nitrogen.
Uracil is referred to as an aromatic compound. Aromaticity is a chemical property in which a conjugated ring of unsaturated bonds, lone pairs, or empty orbitals exhibit a stabilization. This stabilization is stronger than would be expected by the stabilization of conjugation alone. Uracil undergoes amide-imidic acid tautomeric shifts. Tauntomers are isomers of organic compounds that readily interconvert by a chemical reaction, this chemical reaction is called tautomerization. Tautomerism is a case of structural isomerism. The reason this happens to Uracil is because of the molecules' nuclear instability that it may may have from a lack of aromaticity formal that was compensated by the cyclic-amidic stability.
As a pyrimidine nucleobase, Uracil is considered to be a heterocyclic aromatic organic compound. A heterocyclic compound is a cyclic compound that contains atoms of at least two different elements as a member of its ring or rings. The opposite of a heterocyclic compound is a homocyclic compound, in which the ring or rings is made of a single element. Since Uracil is a heterocyclic compound it contains different elements on it's ring, those elements are; carbon and nitrogen.
Uracil is referred to as an aromatic compound. Aromaticity is a chemical property in which a conjugated ring of unsaturated bonds, lone pairs, or empty orbitals exhibit a stabilization. This stabilization is stronger than would be expected by the stabilization of conjugation alone. Uracil undergoes amide-imidic acid tautomeric shifts. Tauntomers are isomers of organic compounds that readily interconvert by a chemical reaction, this chemical reaction is called tautomerization. Tautomerism is a case of structural isomerism. The reason this happens to Uracil is because of the molecules' nuclear instability that it may may have from a lack of aromaticity formal that was compensated by the cyclic-amidic stability.
Uracil's Properties
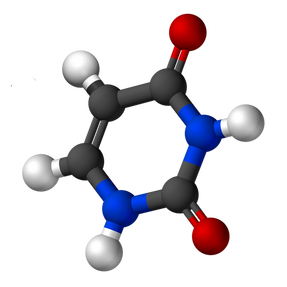
Properties of Uracil
Molecular Formula = C4H4N2O2
Molar Mass = 112.08676 g/mol
Density = 1.32 g/cm3
State = Solid
Boiling Point = N/A (Uracil only decomposes)
Melting Point = 335 Degree Celsius
Solubility = Soluble in Water (H2O)
Flammability = Non-Flammable
Molecular Formula = C4H4N2O2
Molar Mass = 112.08676 g/mol
Density = 1.32 g/cm3
State = Solid
Boiling Point = N/A (Uracil only decomposes)
Melting Point = 335 Degree Celsius
Solubility = Soluble in Water (H2O)
Flammability = Non-Flammable
